Scientists find a glitch in the protocols used to synthesize graphene and 2-D materials

A team of researchers at the Indian Institute of Technology Gandhinagar has stumbled upon an unexpected phenomenon with significant implications for the existing protocols followed to synthesize graphene and other two dimensional (2-D) nanomaterials.
Since its discovery 15 years ago, graphene has led to several breakthroughs in fundamental and applied science. It has also also led to the development of an entire landscape of new 2-D nanomaterials, which are like graphene but made from other elements. One of the most popular methods to synthesize graphene is liquid-phase exfoliation, in which the graphite powder is mixed in a suitable liquid medium and exposed to bursts of high-intensity sound energy (ultrasonication). This ultrasonic energy delaminates the layered parent crystals into daughter nanosheets that suspend and swim in the organic solvents to form a stable dispersion of 2-D nanomaterials; such liquid dispersions are processable and hence very widely used.
In such strategies, it has always been presumed that the ultrasonic energy bursts would not affect the organic solvent. However, the research team at IIT Gandhinagar unintentionally came across some findings that suggested otherwise. They were extending this method to synthesize dispersions of borophene, a 2-D material that is like graphene but made from boron. While conducting the control experiments, Ph.D. student Saroj Kumar Das made an observation that surprised the team. He observed that during this process, the organic solvent itself was transforming into quantum dots − extremely tiny fluorescent nanostructures just two nanometers in diameter, same as the diameter of a DNA molecule.

"We came across this finding unintentionally. When I was shining the dispersion with laser to observe Tyndall effect, I found that the dispersion exhibited beautiful fluorescent colors, a behavior that is characteristic to quantum dots," said Das first author of this study. He reported this unexpected result to his advisor Dr. Kabeer Jasuja.
"Initially, we could not accept the outcome, and we thought that these quantum dots could be arising from a possible contamination. However, after conducting several experiments in carefully designed setups, we were able to identify that the organic solvent used during the process is itself transforming into carbon quantum dots," said Jasuja.
"We were surprised to see this finding, as many scientists, including us, believed that that the liquid medium/ organic solvent is stable during the entire process during exposure to sound waves and nothing happens to it. That is the main reason these are used as a dispersing medium for such experiments in the first place. No one has ever suspected that the molecules of organic solvent can transform into carbon quantum dots by the sound energy," added Das.
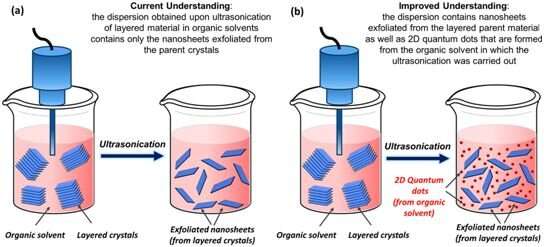
To demonstrate the relevance of these results, the research team went beyond the graphene synthesis protocols and also revisited methods that utilize ultrasonication to synthesize other 2-D materials. "We were able to show that in these protocols, one ends up getting a mixture of carbon quantum dots along with the 2-D nanosheets that are originally intended. These findings imply that before drawing inferences about nanosheets formed by such methods, one needs to acknowledge the presence of these quantum dots," said Ramchandra Gawas, a co-author of the study.
"The resultant carbon quantum dots are photoluminescent and hence have a significant contribution in the optical spectra of the resultant dispersions. We are hopeful that this new physical insight, that we stumbled upon by chance, would form an important addition to the protocols followed to the synthesize 2-D materials," said Dr. Kabeer Jasuja.