Surface decoration of perovskite can make solar cell stable for operation
Solar cells that convert solar energy into electricity are one attractive route for harvesting clean solar energy. The installed capacity of solar panels has exceeded 100 GW in the last year.
Although the cost of silicon solar cells has fallen a bit recently, they are close to their intrinsic cost limit. Developing new photovoltaic technology is a main focus of scientist and engineers, which may further reduce the levelized cost of energy.
Perovskite solar cells with remarkable efficiencies over 25% represent a significant breakthrough. They may cut down the cost of solar cells by up to 30%. However, their poor stability is one drawback preventing practical applications.
About the surface
Conventional silicon devices have put much attention on surfaces of dangling bonds. These dangling bonds can not only trap carriers, but are also active for structural change. For perovskites, the surfaces are defective and much more active than the bulk counterparts. The degradation of perovskite has also been recognized to be from the surface.
This inspires scientists to engineer the perovskite surface with lots of molecules, such as fullerene and thiophene. These configurations have shown to be more stable than the clean surface of perovskites.
The stronger the bonding, the greater the stability
The earliest such molecules are fullerene derivatives (e.g., PCBM) and Lewis bases (e.g., pyridine, thiophene), which weakly interact with perovskite surfaces via Van der Waal or coordination bonds.
Under working conditions, energies from light may change the structure of such decorated surfaces because of their very low bonding energies. In later developments, ionic molecules that fill the charged defect sites via relatively large electrostatic interactions were anchored to the perovskite. These molecules advanced the lifetime of perovskite solar cells.
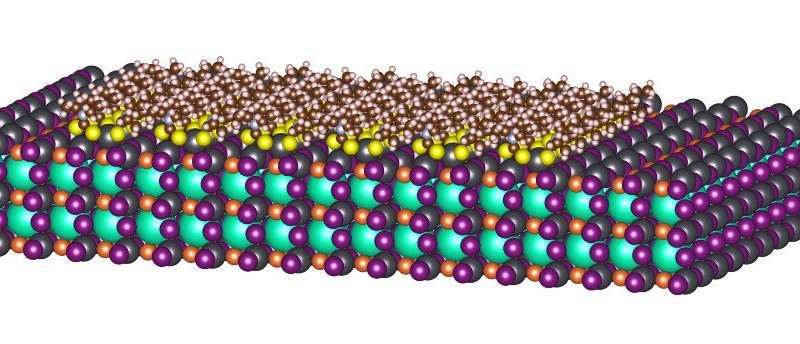
However, it is still challenging to confer even stronger interaction strength to perovskite surfaces that can retain more reliable passivation under various environments. Along this line, we proposed a chelated perovskite surface structure by using dithiocarbamate molecules (Figure shown above).
The surface Pb atoms of perovskite were strongly anchored by two arms form the molecule. This is more stable than molecules with only one functional group.
Device performance
As proved by density functional theory (DFT), the Pb(DDTC)2 has a large adsorption energy of −1.73 eV on the surface, which is five times larger than that of water, at -0.32 eV.
Such strong chemical bonds between the surface with coordinated defects and a chelating agent enables a reliable passivated perovskite surface with long-term stability under various environmental stimuli. Solar cell based on this chelated CsPbI2Br perovskite reaches a record power conversion efficiency of 17.03%, a large open-circuit voltage of 1.37 V. It exhibits excellent stability and can maintain 89% of its initial efficiency for over 520 hours under sun illumination.
These findings provide scientific insights on the surface engineering of perovskites that can facilitate the further development and application of perovskite optoelectronics.
This story is part of Science X Dialog, where researchers can report findings from their published research articles. Visit this page for information about ScienceX Dialog and how to participate.
More information:
Jingjing He et al. Surface chelation of cesium halide perovskite by dithiocarbamate for efficient and stable solar cells, Nature Communications (2020). DOI: 10.1038/s41467-020-18015-5
Dr. Shuang Yang received his bachelor's degree from Tsingtao University of Science & Technology in 2011 and completed his Ph.D. in 2016 at East China University of Science & Technology. He then conducted postdoctoral research at Nanyang and Technological University, University of Nesbraka–Lincoln. Currently, he is a professor at East China University of Science and Technology. His research focus is on the chemistry and physics of semiconducting materials and opto-electrical devices.